- Blog
- Team viewer easy access
- Crane lift plan osha
- Shade 14 eclipse
- Mexican bird of paradise
- Smoke on the water guitar lessons
- Actor arya movie list
- Zimbra outlook for mac zmailcloud
- Freebsd intel gma 4500 graphics
- Hospital umum sarawak
- According to the second law of thermodynamics
- Trina the one album free download mp3
- Jennifer lopez on the floor
- Pes 2017 patch pc
- Mitsubishi ac split unit installation hawaii
- Descargar lumion 9 full
The temperature increase can also be thought of in the following way. There is a law, called the ideal gas law, that neatly describes this process with an equation relating volume, pressure, and temperature of a gas. The compression rapidly increases the temperature of the working fluid. This requires work (and therefore energy) because it forces the fluid to go into a small space. The compressor does what its name suggests: it compresses the gas by application of pressure. The working fluid, which is now a warm, low-pressure gas, circulates through pipes and enters a compressor. But the fluid must deposit this heat outside or the job is not finished. As the working fluid cools, it absorbs heat from the room. According to the first law of thermodynamics, some of their energy is lost. The molecules of the gas lose energy as they expand because they push away the surrounding air molecules, thus doing This cools the fluid in the same way that a breath blown through compressed lips feels cool-although the air comes from the body, which is warm, as the air leaves the compressed lips, it expands. Under low pressure, it evaporates and becomes a gas, which then expands. Chapter 5 describes one that is different but equiva- lent to the above. There are several formulations of the second law of thermo- dynamics. It is not, however, and can never be, because plenty of careful experiments involving heat and work confi rm that the world adheres to the second law of thermodynamics. Getting around the second law of thermodynamics would be terrifi c because then it would be possible to do such things as moving heat from a cold body to a hot one without any energy input. No matter how the attempt is made or what machine or technology is em- ployed, it is not possible to recapture all of the thermal energy and do work with it. Some of this thermal energy can be used to do work, but not all.

Friction and air resistance generates heat, and although the energy of motion that produces this heat is not destroyed, the conversion is not fully reversible.
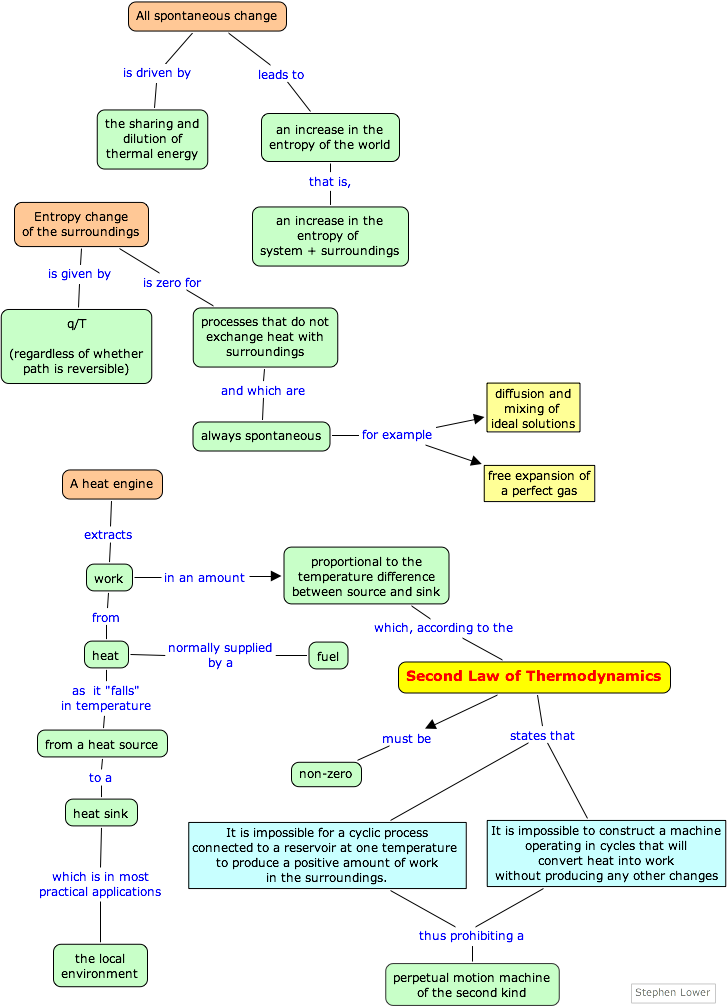
What the second law of thermodynam- ics says is that some of this loss is not reversible.Ĭonsider pushing a cart across a level street, an example discussed in the earlier sidebar, “The First Law of Thermodynam- ics.” Pushing a cart involves work, but unlike lifting a weight, there is little or no energy “stored” in the position of the cart-it cannot return across the street on its own or do work in the process. A lot of heat is generated by friction, for example this is wasteful, for more effort is required in the presence of friction than without it. This is one of the most important constraints of physics. The second law of thermodynamics says that it is impossible to make a machine that functions by completely converting heat, drawn from some body or object at a given temperature, into work (in the sense of physics, where a force moves something over a distance).
